You are here: Nature Science Photography – Image creation, Depth and Size – Photographic image creation
Light transmits visual information about objects, and the Greek word „photography“ means „drawing with light.“ In order to draw a picture that corresponds to our perception, the photographic process must make the information potentials contained in light visible and store them in the same way as our visual system does. It must therefore limit itself to the same area of the spectrum to which we are also sensitive and, first of all, reliably separate black from white and all levels in between.
For this purpose, in analog photography we still rely on the abilities of certain compounds between silver and halogen, fluorine, chlorine, bromine or iodine (silver halide, silver fluoride, silver chloride, silver bromide and silver iodide), which are collectively called silver halides. Although silver is a precious raw material, it has yet to be able to replace it as a direct carrier of image information in analog photography because of its sensitivity to light.
On a chemical level, we make use of the property of silver halides to decompose into their components (e.g., silver and bromine) under the influence of light energy. During this process, the metallic silver turns black, transforming light into a preservable blackness. Since the silver halides in their original form only react to short-wave light (blue and UV), they have to be mixed with other substances to map the spectrum that is visible to us. Dyes such as eosin, fuchsin and cyanine absorb the light energy in the long-wave range of the spectrum and pass it on to the silver halides. Only with their use could film materials be produced that were initially sensitive to all regions of the spectrum except red (orthochromatic) and later to the entire spectrum of electromagnetic radiation visible to us (panchromatic). This laid the foundation for the brightness-correct reproduction of tonal values in black-and-white photography that corresponded to our perception and for color photography based on this.
Based on this foundation, the image formation process for negative and reversal film is identical. The light-sensitive layer of the film is made up of silver halide crystals inside a gelatine shell. These crystals are similar to the rod and cone cells in the retina. Since the size of the crystals determines the sensitivity and graininess of the film material (larger crystals require less light to react but are easier to recognize in the finished image), manufacturers strive to keep them as small as possible. Today, their diameter is between 0.0002 and 0.002 mm. A silver halide crystal consists of about 20 billion silver and halide ions each. An ion is an atom in which the positive charge of the nucleus and the negative charge of the shell do not neutralize each other. The missing or surplus electrons create a positive or negative excess charge. Due to the electromagnetic energy acting at the moment of exposure, the crystals change their charge: A negatively charged halogen ion gives up an electron, which is taken up by a positively charged silver ion. Thus, some silver ions are separated from the halide ions and elemental halogens and metallic silver are formed. Since only a few silver atoms, called development nuclei or exposure nuclei, are formed per crystal during the exposure (their number is usually in the two-digit range), the resulting image is much too faint for us to see. We call it the latent image. It is very durable and the stronger the exposure, the clearer it is. It should be noted here that only two different states can be distinguished – exposed or unexposed – and that basically makes silver films digital image carriers!
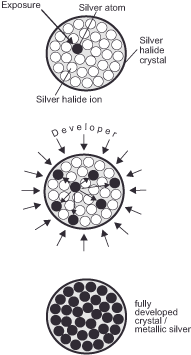
We make the latent image visible by bringing the emulsion into contact with a substance that breaks down all silver halide crystals into the components silver and halogen, but does this more quickly in the exposed than in the unexposed areas. Certain organic substances derived from the benzene ring C6 H6 meet this requirement in combination with special braking agents. We call them developers. They emit electrons that are absorbed by the silver ions and in the process oxidise themselves while absorbing the halides bromine, iodine or chlorine. If the film is developed for the exact time specified by the manufacturer, the special mode of action ensures that only the desired part of the silver halide crystals is reduced. The chapter on „Contrast“ explains how important this is for reproducing brightness values. Interestingly, the border areas between strongly and weakly exposed areas of film are particularly blackened during development, as the developer is used up more quickly in the more strongly exposed areas and can no longer develop the adjacent, less strongly exposed areas evenly. This process, known as the edge effect, increases the sharpness of the image by enhancing the contrasts. – This exciting parallel aligns with our strong orientation towards edges and borders in visual perception, enhancing contrast through the rivalry of adjacent center/surround cells. We place the film into an acidic stop bath, which neutralizes the developer, to safely finish the development.
After this procedure, there are still unexposed and undeveloped silver halide crystals in the emulsion. If we left them there, they would disintegrate into metallic dark silver on their own after sufficiently long contact with light, ruining the image. To prevent this, we wash out the unwanted crystals with a sodium thiosulfate salt bath. We call this process fixing. Afterwards, the unexposed parts of the film are almost transparent.
A long and strong exposure would break down all silver halide into its components over time, resulting in a direct blackening of the layer without any development. This can be observed if we pull the film out of the cartridge bit by bit in daylight. And direct image formation is also possible without development if a panchromatic black-and-white film is exposed for six to eight hours at an open aperture with a sharply focused subject. Once fixed and watered, the film will display an almost normal negative.
Next The negative film
Main Image creation, Depth and Size
Previous Sixth step- Generation of impressions
If you found this post useful and want to support the continuation of my writing without intrusive advertising, please consider supporting. Your assistance goes towards helping make the content on this website even better. If you’d like to make a one-time ‘tip’ and buy me a coffee, I have a Ko-Fi page. Your support means a lot. Thank you!


 Since I started my first website in the year 2000, I’ve written and published ten books in the German language about photographing the amazing natural wonders of the American West, the details of our visual perception and its photography-related counterparts, and tried to shed some light on the immaterial concepts of quantum and chaos. Now all this material becomes freely accessible on this dedicated English website. I hope many of you find answers and inspiration there. My books are on
Since I started my first website in the year 2000, I’ve written and published ten books in the German language about photographing the amazing natural wonders of the American West, the details of our visual perception and its photography-related counterparts, and tried to shed some light on the immaterial concepts of quantum and chaos. Now all this material becomes freely accessible on this dedicated English website. I hope many of you find answers and inspiration there. My books are on